BS, Furman University, 1970; PhD, Oregon State University, 1974
University of Trondheim (Norway), 1975-77; North Dakota State University, 1977-80
General and physical chemistry, instrumental analysis
Asterisks indicate undergraduate student co-authors who conducted their research in collaboration with a faculty member at Colgate.
NSF, Petroleum Research Foundation and Research Corporation grants
My research interest is in the area of gas phase molecular structure determination. I am interested in the accurate measurement of molecular structures using the technique of electron diffraction which involves the computer analysis of diffraction patterns generated by intersecting a beam of electron with a stream of gaseous sample. The analysis is frequently augmented by ab initio molecular orbital calculations.

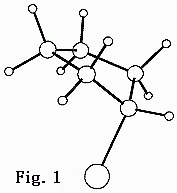
My research efforts have been focused in the following areas: a) cyclic compounds with low frequency torsional motions, b) equilibrium conformational compositions of organic compounds and c) sterically congested molecules.
The non-planarity of cyclopentane and substituted cyclopentanes are known to undergo low frequency pseudorotational motion where the puckered atom moved freely about the ring from one position to another. We have found that substituted cyclopentanes underwent pseudorotational motion with a low energy barrier. We are extending this project to other substituted cyclopentanes and cycloheptanes.


Electron diffraction can be used to study both the structure and the conformation composition of the sample. Different conformations give different diffraction patterns which are recorded as data and careful analyses of these data can resolve the structures and the compositions of the conformers. In addition temperature variation of the composition can be used to calculate thermodynamic quantities like entropy and enthalpy differences between the different rotatomers. We have studied systems like allyl halides, substituted acetones and substituted propenes and butenes.
The structural features of sterically congested molecules can provide insight into the nature of chemical bonding when they are compared with the unstrained systems. We have studied the structures of (E) and (Z)1,2,3 trichloropropenes, and (Z) and (E) ß-bromostyrenes and others.
Year in and year out, I've had the opportunity to teach and interact with capable, intelligent, and motivated students. One of the best collaborative learning environments is Colgate's summer research program where students and faculty work side by side in the laboratory collecting and analyzing data, and developing new models to test hypotheses.